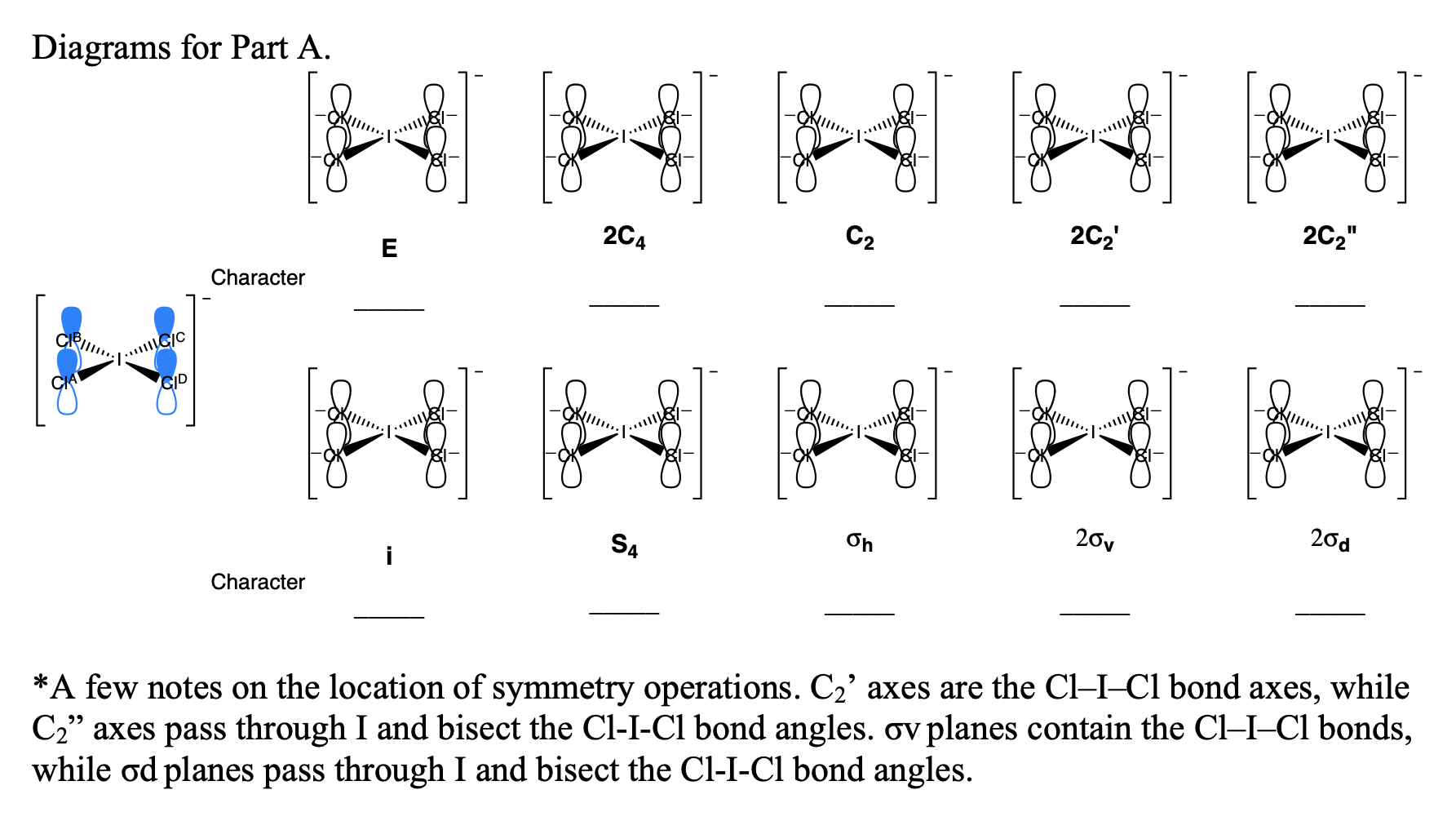
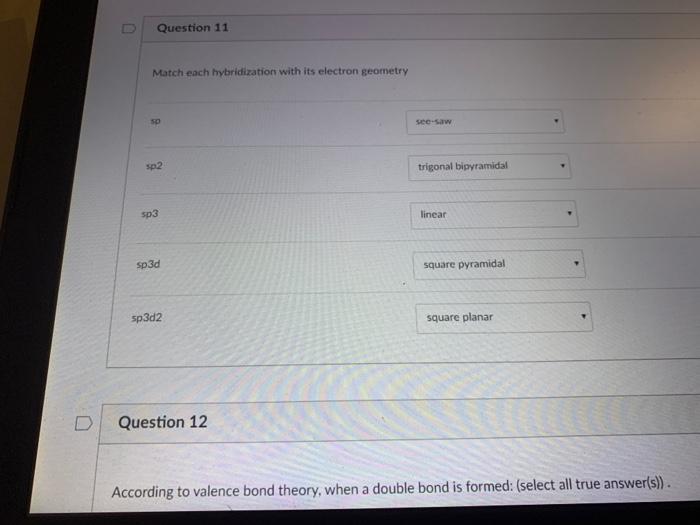
a) Symmetry breaking from octahedral to square planar (left to center)... | Download Scientific Diagram

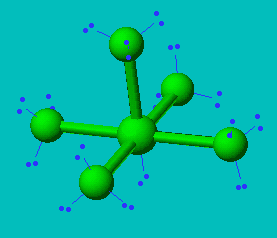
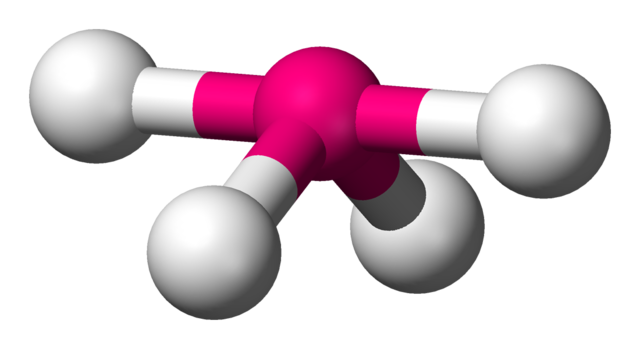
Seesaw Molecular Geometry - Seesaw Shaped Molecules, Lone pairs, Examples & Hybridisation of Seesaw Molecular Geometry
What makes a seesaw-shaped molecule instead of a square planar or tetrahedral? I understand that tetrahedron has 0 lone pairs, but what about the others? - Quora
What makes a seesaw-shaped molecule instead of a square planar or tetrahedral? I understand that tetrahedron has 0 lone pairs, but what about the others? - Quora

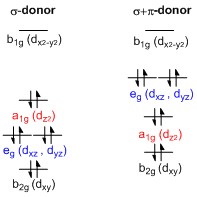
Tetrahedral (left) and square pyramidal (right) distortions for square... | Download Scientific Diagram

Seesaw Molecular Geometry - Seesaw Shaped Molecules, Lone pairs, Examples & Hybridisation of Seesaw Molecular Geometry
What makes a seesaw-shaped molecule instead of a square planar or tetrahedral? I understand that tetrahedron has 0 lone pairs, but what about the others? - Quora
What makes a seesaw-shaped molecule instead of a square planar or tetrahedral? I understand that tetrahedron has 0 lone pairs, but what about the others? - Quora

The molecular geometry of XeO_4^4- is: a. Linear. b. Trigonal planar. c. Seesaw. d. T-shaped. e. Octahedral. f. Square planar. | Homework.Study.com

The molecular geometry of X e F 4 is (a) tetrahedral. (b) seesaw. (c) square planar. (d) square pyramidal. | Homework.Study.com